 > English > Climate Encyclopaedia > Climate in Cities > more > 1. Air Pollution > * Worksheet 1
> English > Climate Encyclopaedia > Climate in Cities > more > 1. Air Pollution > * Worksheet 1
|
|
|
|
|
Climate in citiesRead more |
|
|
|
|
Exhaust gases are dangerous to your health and should not, therefore, be inhaled! Use a fume hood or work outside. You will need: TAKE CARE: Some of the chemicals used in the experiment are harmful. Experiment carefully and use safety equipment!
Preparation of the Saltzmann-Solution. This solution is used to measure nitrous acid (HNO2). This is formed when nitrogen oxides react with water. Dissolve: Basic materials needed for each group (you may also need other other materials, it depends on how you plan your experiment!):
|
1. Preparation and collection of exhaust gas samples in freezer bagsYou will need collection bags to collect and transport your exhaust gas samples. The best bags to use are commercial freezer bags (without a fastener). Before using them as collection bags they have to be modified as follows: Materials needed for each bag:
|
|
|
Place the glass tube in the middle of the open side of the plastic bag. Make sure it reaches well into the bag. Seal the bag by applying tape to the open edges of the bag. Then wrap these sealed edges around the glass tube and tape again tightly (see pictures 1-3 ). Attach the glass stop cock to the end of the glass tube using the bored rubber bung.
|
|
|
|
2. Taking the sample |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||
3. Qualitative analysis of exhaust gases (CO, CO2 and NOx)
Information on the identification methods: 1. Identification of carbon monoxide using an ammoniac solution of silver nitrate: Properties of carbon monoxide: solubility in water 0.23 g L-1 (20°C), boiling point: -191°C, reducing agent, highly toxic in concentrations w>5%.
2. Identification of carbon dioxide with lime water (aqueous solution of calcium hydroxide): Properties of carbon dioxide: solubility in water 1.78 g L-1 (20°C), sublimes at: -79°C.
3. Identification of nitrogen oxides with Saltzmann solution: Properties of nitrogen dioxide: reacts with water yielding HNO2 and HNO3, boiling point:21°C, vapour pressure: 1013 hPa at 20°C, highly toxic in concentrations w>1%.
|
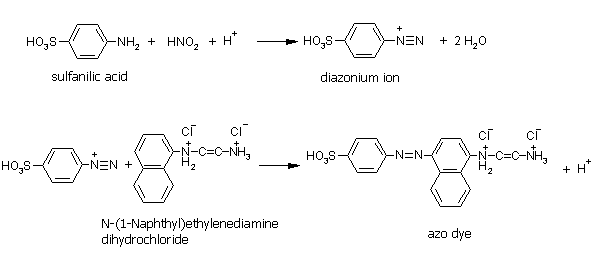 |
|
8. The reaction scheme for nitrous acid identification using the Saltzmann solution. |
|
Exhaust gases contain substances which can damage your health: Carry out the experiments in a fume hood or work outside !
|
About this page:authors: M. Seesing, M. Tausch - Universität Duisburg-Essen, Duisburg, Germany
|







