> English > Climate Encyclopaedia > Upper Atmosphere > basics > 2. Ozone hole > - ozone formation
> English > Climate Encyclopaedia > Upper Atmosphere > basics > 2. Ozone hole > - ozone formation
|
Upper AtmosphereBasics |
Formation of ozoneIf it wasn't for stratospheric ozone, life as we know it now wouldn't be possible on Earth. Ozone prevents harmful ultra-violet radiation from the Sun (light with wavelengths less than 320 nm) reaching the ground. If allowed to reach Earth, this radiation would severly damage the cells that plants and animals are made up of. Ozone was first formed in the Earth's atmosphere after the release of oxygen, between 2000 and 600 million years before the first humans appeared.
|
|
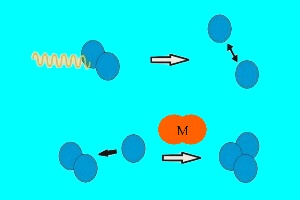
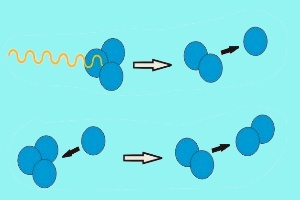
Ozone formation and destructionTwo forms of oxygen are found in the stratosphere. Molecular oxygen (O2), which is made up of two atoms of oxygen (O), and ozone (O3) which, as you can see from its chemical formula, is made up of three oxygen atoms. Ozone is formed when intensive ultra-violet radiation from the Sun breaks down O2 into two oxygen atoms. These highly reactive oxygen atoms can then react with more O2 to form O3.
|
|
In a similar way, ozone is destroyed by solar radiation. Ultraviolet radiation hits ozone and breaks it back down into molecular oxygen (O2) and atomic oxygen (O). The oxygen atom O then reacts with another ozone molecule to form two oxygen molecules. |
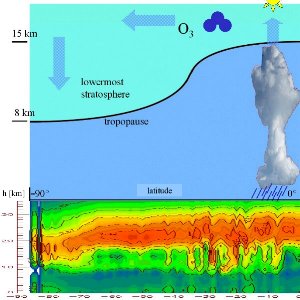
Formation in the tropics, accumulation in polar regionsSince solar radiation is strongest over the tropical regions, most of the ozone is formed in there. The Sun doesn't just drive this tropical ozone formation but also allows tropospheric air to rise to higher altitudes here.
|
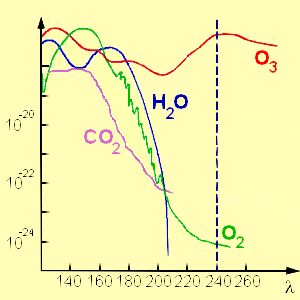
Ozone absorbs ultra-violet radiationAbsorption of the Sun's ultra-violet radiation during ozone formation and destruction in the stratosphere has three important consequences.
|
|
|
Thickness of the ozone layerThe term "ozone layer" is often misunderstood. Its not really a single layer in the atmosphere where ozone is concentrated. Rather it means that a higher fraction of ozone molecules are found in the stratosphere (at altitudes between 18 and 40 km) compared to levels the troposphere below or the mesosphere above. In reality, only about 10 out of every million molecules in the atmosphere are actually ozone but 90% of all the ozone present in the atmosphere is found in the stratosphere.
|
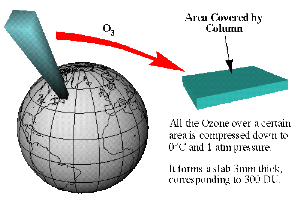
Dobson units (DU)You will often see ozone levels reported in Dobson units (DU). 300 DU is a typical value. But what does this mean? If we assume that all the ozone molecules in the atmosphere were concentrated in a small layer at the ground (rather than being spread over the whole stratosphere and troposphere) then then thickness of this layer would be about 3 mm. Since 1 DU is equivalent to a layer of pure ozone molecules 0.01 mm thick, a 3 mm layer of ozone is equivalent to a value of 300 DU.
|
|
About this page:author: Dr. Elmar Uherek - Max Planck Institute for Chemistry, Mainz, Germany.
|