> English > Climate Encyclopaedia > Lower Atmosphere > more > 3. Ozone & fire > - fire
> English > Climate Encyclopaedia > Lower Atmosphere > more > 3. Ozone & fire > - fire
|
Lower AtmosphereRead more |
Fire chemistry and its global importanceEvery year, large areas of our planet burn:
|
|
10 million hectares of boreal forest in the northern latitudes where 1 million hectares = 10,000 km2 Imagine the area:
|
|
| |||||||
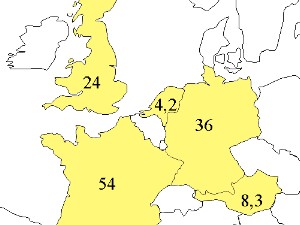
The global carbon budget of CO2 exchange for the period 1850 - 1990.Values in Pg Carbon (Pg C) 1 Pg = 1015 g = 1 billion tons. Emissions from man-made fires are an important source of carbon to the atmosphere. |
 |
|
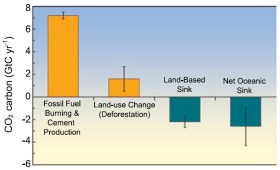
3. Carbon dioxide - sources and sinks - image: Elmar Uherek
|
|
The main sources are:
|
The main sinks are:
|
|
Natural vegetation fires can be short term CO2 sources to the atmosphere. Over the long term, however, natural fires have either very little influence on CO2 levels or may actually represent a very small carbon sink (< 0.1 pG C y-1) since carbon containing sediments are stored in the ground. Open forest and savannah fires occur in cycles of 1-3 years over much larger areas than rainforest fires and have little influence on the climate system. |
 |
|
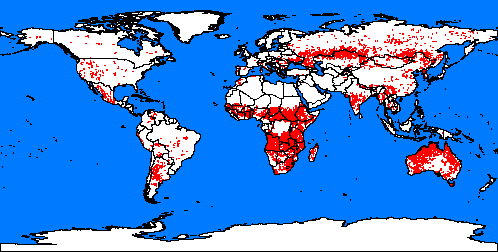
5. Fires spots around the word. Map: from Global fire monitoring centre.
|
What is formed during the combustion process?This depends on; What do plants consist of?Plants contain up to 60% water although their water content is generally lower than this before the fire season as the vegetation looses a lot of water during drought times. The typical elemental composition of the dry organic matter in plants is; Plants typically have the following components:
|
|
|
The burning processAfter ignition, oxygen levels are sufficiently high to allow a period of open fire to occur. As oxygen levels fall, the fire begins to smoulder. During the period of open fire, a simple mixture of fully oxidised compounds, including CO2, are formed. Incomplete oxidation occurs during the smouldering fire stage and a complex mixture of compounds are formed which may even include methane (CH4).
|
|
| |||||||
|
After ignition (> 180°C / 450 K) flaming combustion (> 580°C / 850 K) starts. Simple molecules such as CO2, H2O, NO, N2O, N2 and SO2 are released as oxidation products. As oxygen levels fall, the flames cease .... |
... and smouldering (< 580°C / 850 K) begins. Due to a lack of oxygen, CO and many partially oxygenated organic compounds (including formaldehyde, acetaldehyde, methanol, acetone and methane) are released.
|
|
Related pages Examples of fires and the types of gases released from them are given in:
|
scientific reviewer: Dr. Rolf Sander - Max Planck Institute for Chemistry, Mainz 2004-05-18 last published: 2004-04-06 |