> English > Climate Encyclopaedia > Lower Atmosphere > basics > 3. Ozone and nitrogen oxides > - ozone
> English > Climate Encyclopaedia > Lower Atmosphere > basics > 3. Ozone and nitrogen oxides > - ozone
|
Lower AtmosphereBasics |
Tropospheric OzoneOzone is probably the most famous gas in climate science. Why is this the case?Ozone is a very contradictory gas. It is essential in the stratosphere where it protects us from damaging ultra-violet radiation from the Sun, but it is harmful in the troposphere with high levels causing health problems. In some large cities, car traffic is banned on particular days to prevent ozone smog events from occurring.
|
|
As well as causing health problems, tropospheric ozone acts as a strong greenhouse gas and contributes to global warming. In this unit we look at how tropospheric ozone is harmful to plants and humans and how it acts as a greenhouse gas. In the topic on the Upper Atmosphere, we look at how stratospheric ozone protects us from harmful ultra-violet radiation.
|
|
Please click images to enlarge them! (about 25 KB) |
|
Danger to the respiratory systemOzone is a reactive and irritant gas and, in high concentrations, leads to respiratory problems. It causes inflammation in the lungs and bronchia. Our bodies try their best to protect our lungs from the ozone. However, preventing ozone from entering the lungs also reduces the amount of oxygen we can take up and this makes our hearts work harder. People with respiratory problems such as asthma are particularly at risk of health problems. In the worst case, high ozone levels can cause death.
|
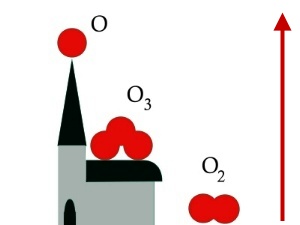
Ozone - a special form of oxygenOzone is a special form of oxygen. Normal oxygen molecules (O2) consists of two oxygen atoms whereas ozone consists of three (O3). It is less stable, more reactive and is able to destroy organic material. This is how it damages plants and causes human health problems. We look at how this happens in more detail in the 'read more' section of this Unit.
|
|
|
Forms of oxygen in the air
ppb = parts per billion
|
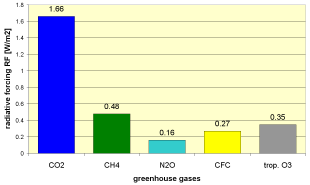
Ozone as a greenhouse gasAfter carbon dioxide (CO2) and methane (CH4), tropospheric ozone is the third most important greenhouse gas, able to absorb heat radiation coming from the surface of the Earth and trap this heat in the troposphere. The additional radiative forcing (
|
|
Calculate how much each greenhouse gas contributes to global warming!The diagram above shows the contribution of several greenhouse gases to the positive change in radiative forcing
Can you calculate the temperature change (
|
|
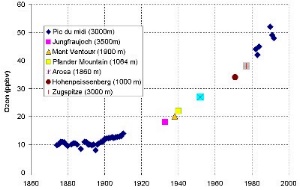
Related pages Find out more about tropospheric ozone in: Background values of tropospheric ozone and their measurements are discussed in the
About this page:author: Dr. Elmar Uherek - Max Planck Institute for Chemistry, Mainz, Germany
|