> ENC Master > Climate Encyclopaedia > Lower Atmosphere > basics > 3. Ozone and nitrogen oxides > - ozone
> ENC Master > Climate Encyclopaedia > Lower Atmosphere > basics > 3. Ozone and nitrogen oxides > - ozone
 |
|
|
|
Lower AtmosphereBasics |
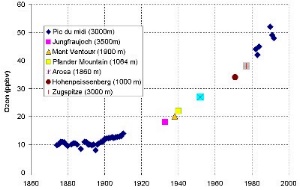
Tropospheric OzoneOzone is probably the most famous gas in climate science. Why?It is a very contradictory gas, sometimes needed, sometimes not. Some people complain, because of the ozone smog. In the worst case you are not allowed to go by car, because too much ozone may be formed. |
|
On the other hand people complain, because the ozone layer becomes thinner and the ozone is urgently needed in order to prevent us from skin cancer. And finally some scientists even tell us, ozone is a greenhouse gas and contributes to Earth warming. Who is wrong???
|
|
source: Innes, Skelly, Schaub - Ozon, Laubholz- und Krautpflanzen, ISBN 3-258-06384-2, Copyright by Haupt Verlag AG / Switzerland Please click images to enlarge them! (about 25 KB) |
|
Danger for the respiratory systemOzone is a reactive and irritant gas and in high concentrations it leads to respiratory problems. It can cause inflammation in the lungs and bronchia, which are usually reversible. Our body tries to protect the alveoli in the respiratory system from ozone. But if less ozone can penetrate, also less oxygen is let in ... the oxygen supply is too low, the heart has to work too much. In particular people, who already have health problems (e.g. asthma), are in danger. In the worst case ozone can be the elicitor of death.
|
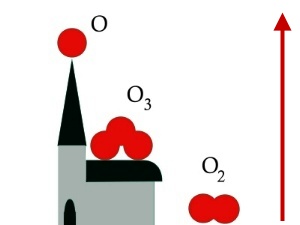
A form of oxygenOzone is a special form of oxygen. Normal oxygen (O2) consists of two oxygen atoms, ozone consists of three (O3), is less stable and more reactive, we could also say aggressive. Ozone is able to destroy organic material as explained for example in 'More - Unit 3'. This is why it attacks the human organism and also the vegetation, as shown in figure 1 in the case of leaves.
|
|
|
Table: forms of oxygen in the air
ppb = parts per billion
|
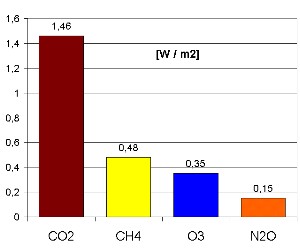
A greenhouse gasFinally, like carbon dioxide (CO2) and methane (CH4), also ozone in the troposphere absorbs the heat radiation coming from the Earth and contributes to the greenhouse effect. It is the third most important greenhouse gas. The additional radiative forcing
|
|
|
Calculate the global warming contribution! The diagramme above shows the contribution of several greenhouse gases to the positive change of the radiative forcing
Can you calculate the temperature change
|
|
Related Pages You will find more information on tropospheric ozone in
|
About this page:author: Dr. Elmar Uherek - Max Planck Institute for Chemistry, Mainz
|