> English > Climate in brief > - Atmosphere
> English > Climate in brief > - Atmosphere
The AtmosphereThe air surrounding us
|
Although you cannot see anything, air is made up of matter. As skydivers show, you can dive in air in the same way as you can dive in water. The resistance is much smaller but it is still there. The air surrounding us consists of countless invisible molecules and atoms floating in empty space. These molecules and atoms interact with radiation from the sun and with each other, starting off chemical reactions. Bigger molecules can also stick to each other and form little particles. Water molecules can condense onto these particles and form cloud droplets. If more and more cloud droplets are formed and grow they finally form clouds which appear in the sky.
|
|
Dimension of the atmosphereLooking into the sky we can easily overestimate the thickness of the atmosphere, it is actually a very thin layer above the Earth. If we travel in a plane at an altitude of between 11 and 12 km (that's between the troposphere and the stratosphere), about 75% of the molecules in the air are below us. This means that an atmospheric layer, less than one thousandth of the Earth's diameter (12800 km), contains three quarters of the mass of the whole atmosphere. In comparison, this is equivalent to a layer of snow less than one centimetre thick on a four storey building. In this layer, clouds form and all our weather takes place.
|
|
|
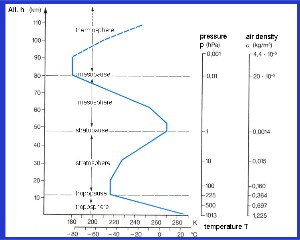
The temperature profile and atmospheric layersWe cannot see the layers in the atmosphere with our eyes but we can measure several changes in the temperature trend as we go from the Earth's surface to higher altitudes. These changes in temperature trends define the atmospheric layers: The troposphere, the lowest layer of the atmosphere, where temperature decreases with height.
|
|
Between the layers there are certain points where the temperature trends change. They are known as '-pauses'. So between the troposphere and the stratosphere we have the tropopause. Above the stratosphere there is the stratopause. The mesopause is found between the mesosphere and the thermosphere and this is the coldest point in the atmosphere. Temperatures here can be as low as -100 oC! In contrast to the temperature, the pressure and density of the air decreases continuously with altitude as the air gets thinner. For a fixed volume of air, there will be 1000 oxygen molecules near the surface of the Earth but at an altitude of 50 km, the same volume of air will contain just one oxygen molecule.
|
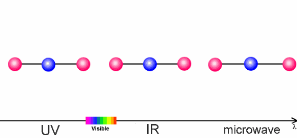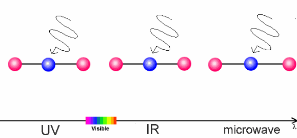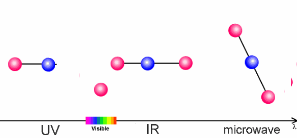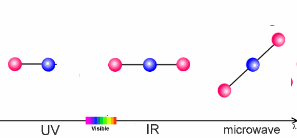
The interaction of light and airThe sun warms the surface of the Earth and, as a result, the further away we are from the surface, the lower the temperature. This process is what controls the temperature trend in the troposphere, the lowest layer of our atmosphere. What causes this temperature trend to change direction in the stratosphere? The air becomes warmer if air molecules can absorb energy from the sun themselves. In the stratosphere it is the ozone molecules of the ozone layer which take up the energy. The properties of such molecules are extremely important to our climate. The amount of energy a molecule absorbs depends on the molecule itself and on the wavelength (energy) of the light.
|
The animation on the right shows three examples:
|
|
|
The absorption of energy from the Sun by ozone in the stratosphere keeps the Sun's energy in this layer and is the reason why the temperature of the stratosphere increases with altitude. This is similar to the process which occurs in the thermosphere but here it is oxygen and nitrogen which absorb even more energetic radiation from the Sun. The radiation is so energetic that it does not only break bonds but forms charged atoms and molecules known as ions. This is why the layer is also known as the ionosphere.
|
|
English proof reading: Lucinda Spokes, UEA Norwich - Sally Taylor, Univ. of Leeds last published: 2005-06-14 |