> English > Climate in brief > - Stratosphere
> English > Climate in brief > - Stratosphere
|
|
|
Only chemicals with very long atmospheric lifetimes can reach the stratosphere. However, once in the stratosphere they can remain there for a long time. Material emitted from large volcanic eruptions, such as El Chichon in 1982 and Mount Pinatubo in 1991, stayed in the stratosphere for up to two years. AviationAeroplanes generally fly between 10 and 12 kilometres in altitude. Increased aviation traffic has, therefore, led to higher emissions of carbon dioxide (CO2), water vapour (H2O), nitrogen oxides (NOx), sulphur oxides (SOx) and soot carbon into the atmosphere between the upper troposphere and the lower stratosphere.
|
At the moment planes account for about 2-3% of global greenhouse gas emissions and this is predicted to rise to 3-4% of total emissions in the future. What is really important is that they emit the gases high up in the atmosphere. The water vapour released increases the likelihood that high cirrus clouds will form. These trap heat from the Earth and so encourage global warming. Nitrogen oxides released from aeroplane exhausts are also important to our climate, these are involved in stratospheric ozone depletion.
|
|
|
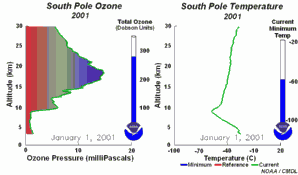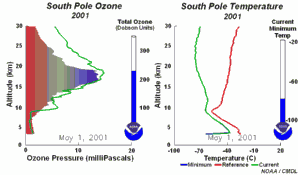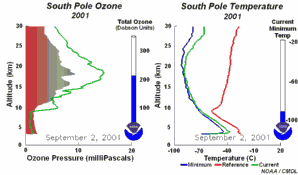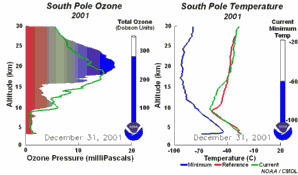
The importance of the ozone layer.Ultraviolet radiation (with wavelengths between 220-320 nm) reaches the Earth and can cause skin cancer if doses are high enough. Fortunately there are high concentrations of ozone in the stratosphere at altitudes between 20 and 45 kilometres. Much of the ultraviolet radiation from the Sun is absorbed by this ozone layer and converted to heat. This protects us from the damaging rays of the Sun.
|
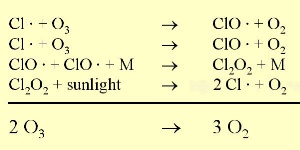
The ozone holeIn the stratosphere, formation and degradation of ozone takes place at the same time. Ozone is formed when sunlight breaks an oxygen molecule (O2) into two oxygen atoms (O). One of the free oxygen atoms then reacts with another oxygen molecule to form ozone (O3). Ozone is naturally broken down by light back into oxygen atoms (O) and oxygen molecules (O2). The oxygen atom then reacts with another molecule of ozone to form two oxygen molecules. This natural cycle of ozone formation and depletion used to be in balance and ozone concentrations in the stratosphere stayed relatively constant.
|
However, there is an another way by which ozone is lost in the stratosphere. Chloroflurocarbons (CFC's) are very stable in the troposphere and can, therefore, reach the stratosphere. Once in the stratosphere they are broken down by ultraviolet light into highly reactive chlorine radicals. These destroy ozone. The same occurs with bromine radicals even more efficiently. Unfortunately, due to human acitivity, high amounts of chlorine and bromine containing compounds such as CFC's have been transported to the stratosphere.
|
|
|
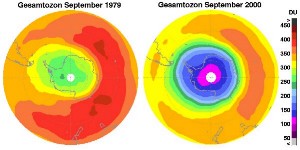
This extra ozone destruction mechanism means that ozone levels have fallen in the stratosphere. Because these extra reactions need light and the formation of polar clouds at very low temperatures, ozone levels are lowest in the Antarctic spring and an ozone hole forms in particular over the Antarctic continent. However, low ozone levels have also been seen over the Arctic. These ozone holes mean that more ultraviolet radiation from the Sun now reaches the surface of the Earth. Human health suffers as a result of the increased incidence of skin cancer and plants are also damaged.
|
|
Have a look at the section on the UPPER ATMOSPHERE in the Climate Encyclopaedia to find out more about the layers in our atmosphere and the ozone layer in particular.
English proof reading: Lucinda Spokes, UEA Norwich - Sally Taylor, Univ. of Leeds last published: 2005-06-14 |