 > ACCENT en > Nr 6 Feb. 2006 acid rain > C: The sulphur cycle
> ACCENT en > Nr 6 Feb. 2006 acid rain > C: The sulphur cycle
|
Context 2: The sulphur cycle
|
|
We find many sulphur compounds on Earth. These include sulphur dioxide, elemental sulphur, sulphuric acid, salts of sulphate or organic sulphur compounds such as dimethylsulphide and even amino acids in our body. All these chemical compounds do not last forever. They are transported by physical processes like wind or erosion by water, by geological events like volcano eruptions or by biological activity. They are also transformed by chemical reactions. But nothing is lost. Changes often take place in cycles. Such cycles can be chemical cycles in which a sulphur compound A reacts to form B, B to C, C to D and D to A again. At the same time there are spatial / geographical cycles. One example is when sulphur compounds move from the ocean to the atmosphere, are transported to the land, come down with the rain and are transported by rivers to the ocean again.
|
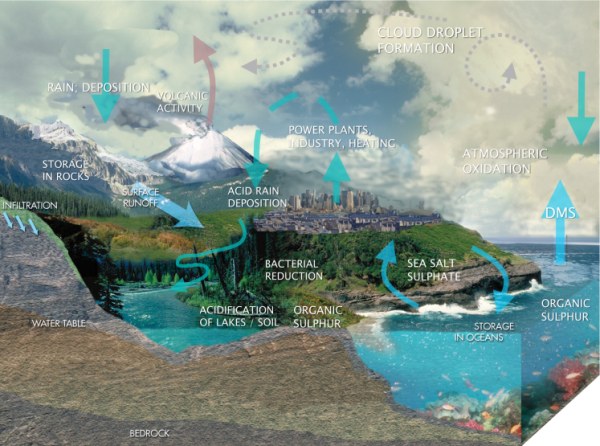 |
|
1. The flow of sulphur compounds in our environment.
|
|
In chemical cycles, sulphur is usually oxidised in the air from organic sulphur or elemental sulphur to sulphur oxides like SO2 and SO3 ending up as sulphate in sulphate salts M(II)SO4, M(I)2SO4 or sulphuric acid H2SO4. The sulphate compounds dissolve very well in water and come down again with the rain, either as salts or as acid rain. In chemical cycles oxidised compounds must also be reduced again. This process does not take place in the atmosphere but on the ground and in the oceans and is carried out in complicated chemical reactions by bacteria. The most important products are elemental sulphur, hydrogen sulphide (H2S), which smells awful and is very unhealthy, and organic sulphur compounds.
|
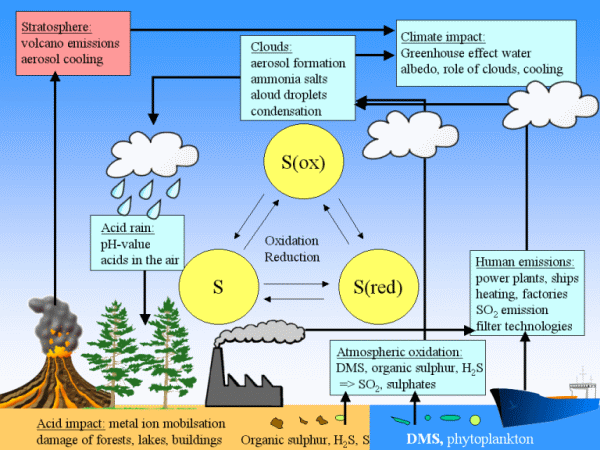 |
|
3. Sulphur compounds play a role in our climate system in various ways. The scheme gives an overview. Click on it for a larger view. (90 K)
|
|

