> ENC Master > Climate Encyclopaedia > Upper Atmosphere > basics > 2. Ozone hole > - ozone formation
> ENC Master > Climate Encyclopaedia > Upper Atmosphere > basics > 2. Ozone hole > - ozone formation
 |
|
|
|
Higher AtmosphereBasics |
Formation of ozoneThe biosphere, the animals on our planet and certainly also human life became possible due to the formation of ozone in the stratosphere. Ozone protects us from the UV-C and UV-B radiation of the sun (light with wavelength of less than 320 nm), which damages the biomolecules. In Earth's history ozone could form after the release of oxygen to the atmosphere between 2000 and 600 Mio before our time. But how?
|
|
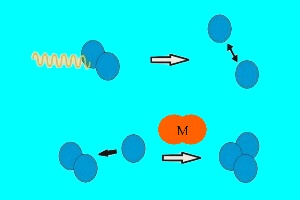
UV light drives ozone formation and destructionIn the stratosphere there are two forms of oxygen: normal oxygen O2 consisting of two O atoms and ozone O3 consisting of three O atoms. In order to transform one form into the other intensive UV-light is necessary. The O-O bond of an oxygen molecule is broken. The formed O atom reacts with oxygen (and for energetic reasons a collision partner M) and forms ozone. This UV-light (yellow wave in the image on the left) comes from the sun.
|
|
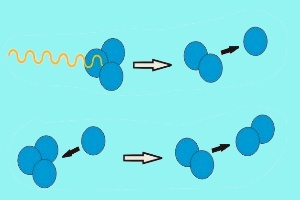
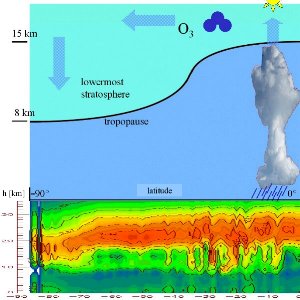
In an analogous way, ozone is destroyed by photolysis, if the O-O bond in an ozone molecules is split by sunlight. In this case the formed O atom reacts with another ozone molecule and forms two oxygen molecules O2. Formation in the tropics, accumulation in polar regionsSince solar radiation is strongest over the tropics, here most of the global ozone is formed. However, the sun in the tropics does not only drive ozone formation, but also the rise of tropospheric air to high altitudes. |
|
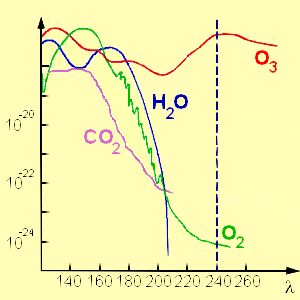
Ozone absorbs UV lightThe same UV light which is necessary for the ozone formation is also absorbed by the molecules of the ozone layer. This has three consequences: 1) This light does not reach the lower parts of the atmosphere and therefore the Earth surface is protected.
|
Thickness of the ozone layerThe term ozone layer is often misunderstood. Ozone layer means, that compared to the troposphere below and the mesosphere above a higher fraction of ozone molecules is found in the stratosphere between 18 and 40 km of altitude. However this fraction is certainly very small compared to nitrogen and oxygen. Not more than 10 of 1 million molecules are ozone. But since ozone in the troposphere is even less and 90% of all atmospheric ozone is in the stratosphere, we call this region the ozone layer. This does not mean that there is somewhere in the atmosphere a thin film of ozone.
|
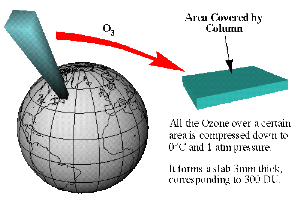
Dobson units (DU)However, ozone is often measured in Dobson Units (DU). 300 DU is a typical value. What does it mean? If we assume that all ozone molecules would not be spread over the whole stratosphere and 10% also over the troposphere, but concentrated in a small film at the ground, the thickness of this film would be about 3 mm (= 300 DU). 1 DU = 0.01 mm film thickness of pure ozone at the ground.
|
|
About this page:author: Dr. Elmar Uherek - Max Planck Institute for Chemistry, Mainz
|