> ENC Master > Climate Encyclopaedia > Upper Atmosphere > basics > 2. Ozone hole > - ozone hole & CFC
> ENC Master > Climate Encyclopaedia > Upper Atmosphere > basics > 2. Ozone hole > - ozone hole & CFC
 |
|
|
|
Higher AtmosphereBasics |
Chlorofluorocarbons CFC and ozone holeThe story of the ozone hole is a good example of how an apparently harmless class of chemical compounds can become a real danger for life on Earth, and how the governments, industry and civil society can work together to identify this problem and act to resolve it. |
|
From this we can learn, that every change man causes in the climate system, may disturb the natural equilibrium in an unexpected way and that if the world community can work together in targetted way, the global environmental problems can be effectively dealt with.
|
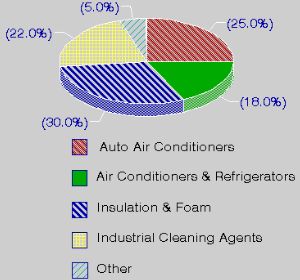
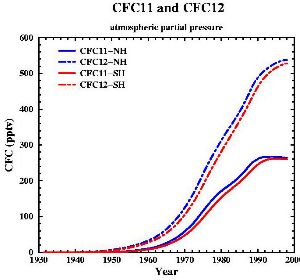
Use and properties of CFCsChlorofluorocarbons are fully halogenated carbon compounds as CFCl3 or CF2Cl2 (commercially they are well known as FREON, although it is a brand name). CFCs have been used in a wide variety of manufacturing steps and products including as a refrigerant, solvent in the electronics industry, foaming or blowing agent, aerosol propellant, fire extinguisher agent, dry cleaning solvent, degreasing agent, a key component in making rigid foam insulation for houses and household appliances, and foam packaging insulation material.
|
|
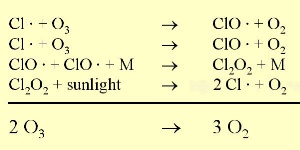
The chemical reactionOzone is in a state of equilibrium in the ozone layer, formed and decomposed due to UV-light. Chlorine radicals (Cl |
|
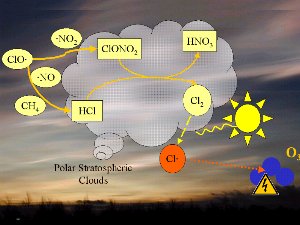
The conditionsThe decomposition of the CFCs leads after further reaction to ClO
|
|
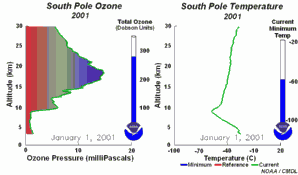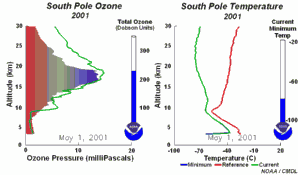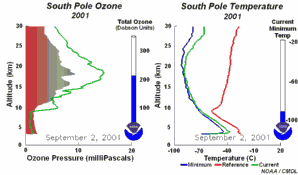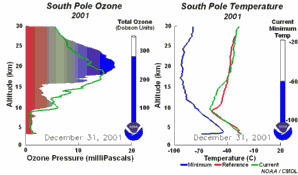
1) One factor is the extremely low stratospheric temperatures of about -80°C or less over Antarctica during the polar night. During such conditions nitric acid and water form stratospheric ice clouds, which are not stable at higher temperatures. On the surface of these ice clouds HCl and ClONO2 react with each other and form nitric acid and pure chlorine Cl2. 2) Chlorine Cl2 is a stable molecule and does not react with ozone but it is easily photolysed by sunlight and forms 2 Cl
|
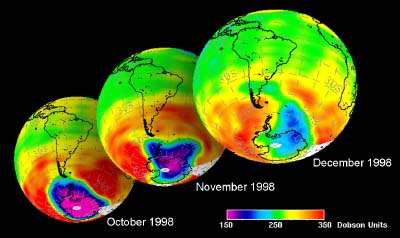
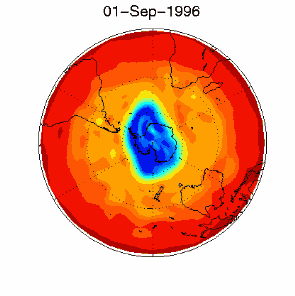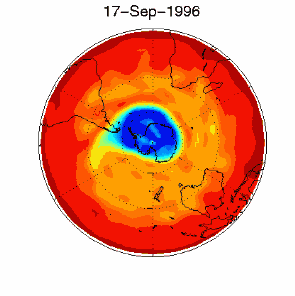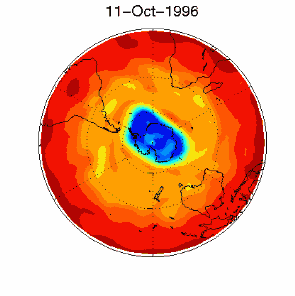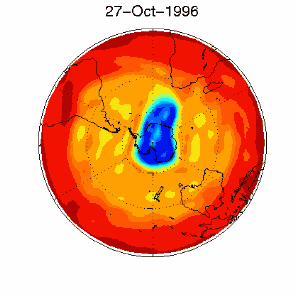
As we saw above, it is the chlorine radical Cl which leads to the ozone depletion. This does not happen before the sunlight for the cleavage is available, i.e. in Antarctic spring. Therefore we observe the ozone hole every year at the same time in September and October. The reaction chain goes as long as also the other reactants in the ice clouds are thawed again and Cl
|
|
|
3) Finally the whole process should not be that dramatic and not affect the most concentrated part of the polar ozone layer between 14-22 km altitude, because the source compound ClO As we see, the conditions for the ozone hole (deep temperatures in polar night, ice cloud formation, polar vortex, followed by the polar sun rise) are that special that this process would have never been predicted by scientists if it would not have been observed.
|
The future of the ozone holeCFCs are meanwhile banned on a global scale (Montreal Protocol on substances that deplete the Ozone layer-1987 and further amendments). Due to their long lifetime it will take about another 50 years until the CFCs released so far are removed again in the stratosphere and the ozone equilibrium is hopefully stable again. It is assumed that around 2000 the maximum has been reached and the ozone hole during the last years was rather stable in its size. However, exceptions are always possible. In 2002 there has been no significant ozone hole observed. The reason was simple: It was too warm and the polar vortex has not been formed in the usual way. Once again an example that atmospheric processes sometimes ignore any prediction. But in 2003 the hole was back to its former size, the second largest ever observed.
|
|
About this page:author: Elmar Uherek - Max Planck Institute, Mainz
|
Recommended reading:
|