> ENC Master > Climate Encyclopaedia > Climate in Cities > more > 3. Acid Rain > - What can we do?
> ENC Master > Climate Encyclopaedia > Climate in Cities > more > 3. Acid Rain > - What can we do?
 |
|
|
|
Urban ClimateRead more |
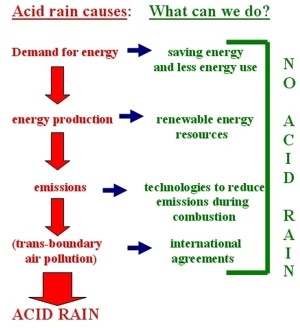
What can we do to counteract acid rain?Each of us can do something to limit the production of acid rain or help neutralise its effects. These actions vary from international agreements, through soil and water liming, to filtering the exhaust gases.
|
|
Two directions All efforts undertaken to counteract acid rain may be divided into two main groups:
|
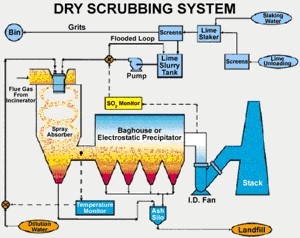
Better technology There are many technologies which can be used in industry to reduce the emissions of pollutants to the atmosphere and these can be applied before, during or after combustion.
|
|
|
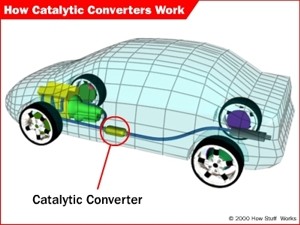
Cars which are equipped with catalytic converters have reduced emissions of oxides of nitrogen. These gases are oxidised in the atmosphere to form acidic rain. Figures 3 and 4 explain how a catalytic converter works. |
|
|
|
International co-operation By the late 1970s concern led to international efforts to identify the causes and effects of long-range (transboundary) transport of air pollutants, and thus during the 1980s much research was conducted in Europe and North America. International legislation during the 1980s and 1990s has led to reductions in SO2 emissions in many countries but reductions in emissions of NOx have been much less.
|
|
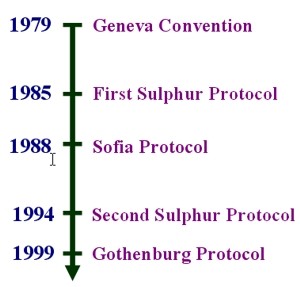
In 1979, the United Nations Economic Commission for Europe (UNECE) implemented the Convention on Long-Range Transboundary Pollution (the so-called Geneva Convention). In 1985, in Helsinki, most UNECE members adopted the Protocol on the Reduction of Sulfur Emissions, agreeing to reduce SO2 emissions by 30% (from 1980 levels) by 1993. This was called the 30% club. All of the countries that signed the Protocol achieved this reduction, and many of those that did not sign, have met these reductions.
|
|
The Sofia Protocol for reducing NOx emissions was adopted in 1988. The emissions should be stabilised against 1987 levels. However, many countries are unlikely to meet these targets, due to increases in road traffic, despite European Union legislation requiring cars built after 1993 to be fitted with a catalytic converter.
In June 1994, a number of European countries signed the Second Protocol for sulfur. Its main objective was reducing acidifying emissions to a level at which critical loads are not exceeded, i.e. reduce sulfur emissions by 70-80% by the year 2000 (against 1980 levels). Eastern European countries generally have a lower target of 40-50% (against 1980 levels).
The most recent UNECE Convention on Long Range Transboundary Air Pollution protocol was signed by 27 countries in December 1999. The Gothenburg Protocol, designed to Abate Acidification, Eutrophication and Ground-level Ozone aims to cut emissions of four pollutants: SO2, NOx, VOCs and NH3, by setting country-by-country emission ceilings to be achieved by the year 2010.
|
|
Neutralising the effects
|
|
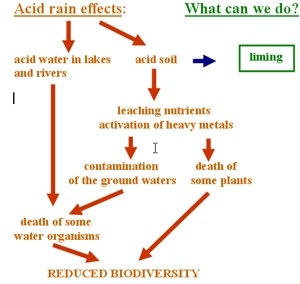
Once the acid rain occurs, all we can do is neutralising its effects. However, as shown in figure 6, our actions can be efficient only at the beginning. When the next stages of the influence of acid rain have already occurred, it is much more difficult to counteract the negative effects. The most commonly used method is liming, described below. |
|
Natural and artificial buffers The resulting acidity of water is greatly influenced by the amount of limestone in the surrounding rock formations. The river beds and lake beds formed from limestone rocks allow the water to dissolve some of it and this neutralizes the acid rain. At present, the main way of artificial reversing acidification in freshwaters is liming the water body or its surrounding catchment, i.e. adding lye (NaOH(aq), sodium hydroxide) or slaked lime (Ca(OH)2(aq), calcium hydroxide). However, it causes aluminum and other metals to come out of solution and fall to the bottom of the lake, improving the conditions for fish life, but causing toxicity problems for organisms living on the lake bed. Acidified lakes in Sweden have been restored in the short term by liming. The same method is used for lakes in USA and Canada. Liming on such a large scale, however, is expensive. Liming provides only a temporary solution, hence it is far better to attack the source of the problem by reducing emissions of acidifying pollutants, SO2 and NOx.
|
|
Related pages: Learn more about the origin of acid rain and double role of ammonia in:
|
About this page:Author: Anita Bokwa - Jagiellonian University - Cracow / Poland |