> ENC Master > Climate Encyclopaedia > Lower Atmosphere > more > 4. Gases in the atmosphere > - Distribution & concentration (1)
> ENC Master > Climate Encyclopaedia > Lower Atmosphere > more > 4. Gases in the atmosphere > - Distribution & concentration (1)
 |
|
|
|
Lower AtmosphereRead more |
Distribution & concentration (1)We learned a lot about the gases in our atmosphere, in particular in the troposphere, the lowest layer of the atmosphere. Here we find countless chemical compounds. But concentrations and distributions are strongly varying.
|
How to decribe a gas in the atmosphere?Amount A gas in the atmosphere can be: a) a major component of the air (oxygen, nitrogen, argon) Trace gases are gases which make up only for a tiny fraction of the air, this can be less than one molecule among one billion or even one trillion of air molecules.
|
|
*The misunderstanding of mixing ratio 'units' ppm and ppbIn numerous scientific publication the amount of a compound in the air is given in ppm (parts per million) or ppb (parts per billion). We also use this abbreviation here, because it is so common and you find it everywhere. But they are very misleading. Three typical mistakes: 1) Often it is written: The concentration of CO2 in the air is 370 ppm. This is wrong. 370 ppm is a mixing ratio and not a concentration. Concentrations are for example mass per volume like the ozone concentration 100 µg / m3. They have a real unit. 2) Mixing ratios do not really have a unit. Because you can cancel the units. They describe only that you have 370 molecules among 1,000,000 molecules, if you have 370 ppm. The more correct expression is: 370 µmol/mol. 3) The term billion and trillion is defined in different ways in different countries:
|
DistributionDepending on local and temporary conditions, a gas in the atmosphere can be:
|
|
|
|
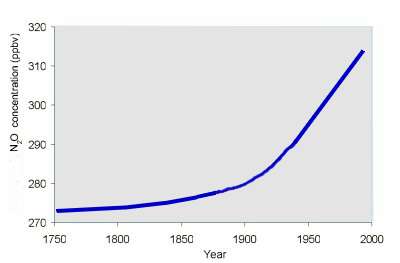
Homogeneously means, that we find comparable mixing ratios of the gas everywhere around the globe and also for varying altitudes. This is the case for stable gases with a long life time. They are only slowly removed from the atmosphere and do not react or only in minor amounts. Example: Nitrous oxide N2O |
 |
|
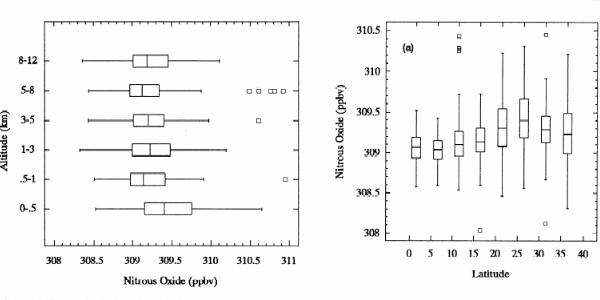
2. Distribution of nitrous oxide in the atmosphere. Measured over the pacific ocean in different altitudes and latitudes. Error bars are shown.
|
Nitrous oxide N2O is an homogeneously distributed gas. However, the concentration has been increasing during the recent 200 years mainly due to human activity. 3. Graph: Elmar Uherek |
|
|
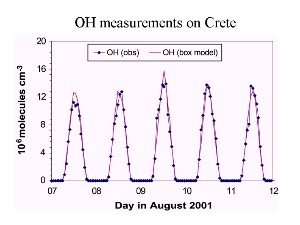
Development in timeThe abundance of gases can strongly depend on the sun, if they are involved in chemical processes, where photolysis plays a role. In this case they have a daily pattern and sometimes also a seasonal pattern. Daily variation: example hydroxyl radical OH OH, for example depends on the sunlight, rises during daytime and decreases at night, es decribed in the unit oxidation.
|
|
Some gases increase or decrease in the long term over decades, hundreds, thousands or even millions of years. Human activity caused an average increase of many gases during the last 200 years after beginning industrialisation as already shown for N2O.
|
|
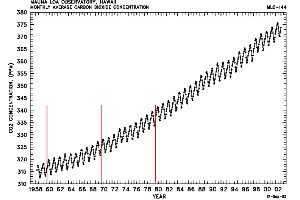
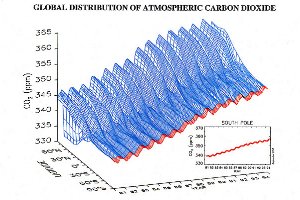
Growing season and human impact: example carbon dioxide (CO2) Carbon dioxide is an excellent example to study the global distribution of a gas. It is rather stable and therefore distributes over the whole globe. But we know: Due to human activity carbon dioxide is continuously increasing over the years, as shown on the left.
|
|
Therefore carbon dioxide increases first in the northern hemisphere and after that slowly finds its way to the south. The transport over the equator takes time, since mixing within one hemisphere is faster than mixing between the hemispheres.
|
About this page:author: Dr. Elmar Uherek - Max Planck Institute for Chemistry, Mainz
|