> ENC Master > Climate Encyclopaedia > Lower Atmosphere > more > 4. Gases in the atmosphere > - Distribution & concentration (2)
> ENC Master > Climate Encyclopaedia > Lower Atmosphere > more > 4. Gases in the atmosphere > - Distribution & concentration (2)
 |
|
|
|
Lower AtmosphereRead more |
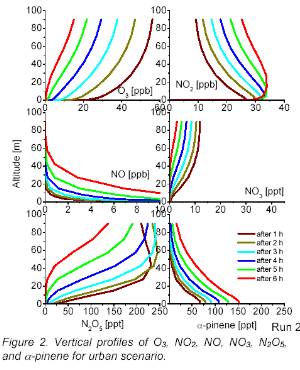
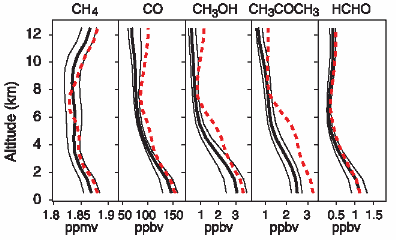
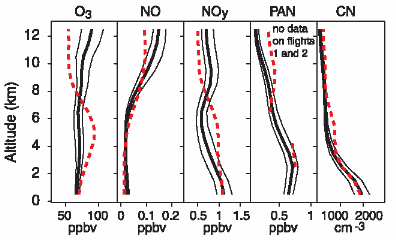
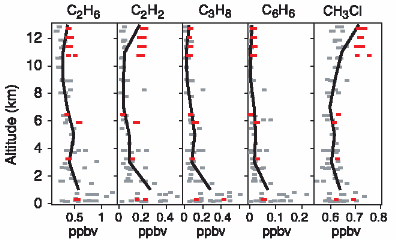
Distribution & concentration (2)If we would like to know about concentrations of less stable compounds in the atmosphere, we cannot just give a number. They depend very much on the chemistry going on in the air.
|
|
Measured compounds: CH4 = methane
|
|
O3 = ozone
|
|
C2H6 = ethane
|
Gases in the troposphereTo give an overview of trace gases in the troposphere and their concentrations is hardly possible. The same compound can be present in extremely low concentrations for example over the ocean and in very high concentrations in the urban environment. Also, a few dozens of gases could be regarded as important. Therefore the following table can only be a compilation of examples, which gives a reasonable range of mixing ratios (usually near the ground) for often measured compounds.
|
|
Overview of important gases in the free atmosphere:
*Gases with increasing concentration due to human influence which are relatively well mixed over the globe. Data from 2001-2003.
|
Mixing ratios, concentrations and different units:Amounts of gases are often given in different units: concentrations: molecules/cm3 or µg/m3 Mixing ratios are often more helpful for scientist, because if air rises and expands the volume grows and therefore the concentration change, but the mixing ratio (relation of the gases) remains the same. 1 mol = 22.4 L = 6x1023 molecules => Example for a rough estimate: So the mixing ratio is about 2.7x1016 / 2.7x1025 = 10-9 = 1 ppb Since ozone has a similar molecular weight, M(O3) = 48 g/mol, we can also roughly say
|
|
Related pages You will find more explanations about concentrations and mixing ratios in
|
About this page:author: Dr. Elmar Uherek - Max Planck Institute for Chemistry Mainz
|