|
|
 |
|
|
|
 |
| |
|
|
 |
Climate in cities
Read more |
Worksheet 1
Air contaminants from exhaust gases of motor vehicles
This worksheet may help you to find out something about the content of exhaust gases as well as the effect of catalytic emission reduction of exhaust gases.
|
|
|
|
|
 |
 |
 |
Picture 8
© FreeFoto.com |
|
 |
 |
 |
Picture 9
© FreeFoto.com |
|
Exhaust gases are hazardous to your health and should therefore not be inhaled!
If possible use a fume hood or work outside.
Some useful information for the experiments:
Chemicals you need:
Be careful: Some chemicals used in the experiment are harmful. Experiment carefully and protect yourselves!
| |
concentrated ammonia (25%) [C, N; R: 34-50; S: 26-36/37/39-45-61] |
| |
aqueous solution of silver nitrate, AgNO3 (aq), (ca. 2%) |
| |
aqueous solution of calcium hydroxide, CaOH (aq) |
| |
N(1-naphthyl)ethylenediamine dihydrochloride [Xi; R: 36/38; S: 22] |
| |
sulfanilic acid [Xi; R: 36/38-43; S: 2-24-37] |
| |
acetic acid [C; R: 10-35; S: 1/2-23-26-45] |
| |
dest. water |
Preparation of the Saltzmann-solution (detection agent for nitrogen oxides, that form nitrous acid with water): 5g sulfanilic acid, 0,050g N(1-naphthyl)ethylenediamine dihydrochloride and 50ml acetic acid are dissolved in 800ml water. The solution is then filled up to 1L. In the absence of light and air the solution can be stored for months.
Materials needed for each group (depending on the pupils´ planning there might be other or more materials needed as well ):
|
3 |
gas-washing bottles
or alternatively: test tubes with lateral tube with offset extension, gas-entry tube (bent glass tube) and suitable plug |
|
2 |
cuvets or small, clean test tubes of the same shape |
|
2 |
glass syringe (with clip) |
| |
retort stands, clamps, sockets |
| |
flexible tube pieces as connecting pieces |
| |
bag for sample collection (see worksheet below, alternatively you can take old plastic
storage bags for wine from Tetrapacks which have to be retrofitted accordingly.) |
| |
flexible tube fitting the gas-entry-tube and the glass stopcock |
|
1. Preparation and sample collection of exhaust gas in freezer bags
You need sample collection bag in order to collect and transport your samples of exhaust gas. You can take different kind of bags but we propose commercial freezer bags (without fastener). Before using them as collection bags they have to be modified as follows: materials needed for each bag:
|
1 |
freezer bag (or a thick airtight and gas-impermeable plastic bag) with a volume of 3 to 10 liters |
|
1 |
tube (glass, plastic or metal) length: 15-30 cm, diameter: 25- 50 mm |
| |
broad plastic tape |
|
1 |
rubber stopper (for the connection of the glass stopcock with the glass tube, see pictures 1-4) |
|
1 |
glass stopcock (or glass tube with rubber hose and pinchcock) |
|
 |
 |
Picture 1
© 2003 M.Seesing |
|
 |
 |
 |
Picture 2
© 2003 M.Seesing |
|
The glass tube should be placed at the middle of the open side of the plastic bag and should reach far into the bag. In order to seal the bag, apply tape first to the sides of the bag, wrap the sides around the glass tube and tape again tightly (see pictures 1-3 ).
|
 |
 |
 |
|
Picture 3
© 2003 M.Seesing
|
|
|
 |
 |
 |
|
Picture 4
© 2003 M.Seesing
|
|
 |
 |
|
Picture 5
© 2003 M.Seesing
|
|
 |
Try to avoid inhaling the exhaust gases!
You can usually take the sample by removing the stopper, wrapping the end of the glass tube with a piece of cloth, removing all air inside the bag, placing the glass tube tightly at the end of the exhaust pipe (see picture no. 5). "Wash" the bag once with exhaust gas before taking the actual sample. As soon as the bag is filled with gas for a second time attach the stopper to the glass tube, c.f. picture no. 4 (after having closed the pinchcock). |
 |
 |
|
Picture 6
© 2003 M.Seesing
|
|
 |
If the procedure decribed above is not possible proceed as follows: Take a glass tube and attach a cork stopper to one end. The stopper should also fit the vent of the exhaust pipe. The other end of the glass tube is connected to the glass tube of the collecting device via a rubber hose (see picture no. 6). For this alternative a greater gas pressure is needed. Therefore it is necessary to close all vents which are connected to the vent where the sample is taken with cork stoppers. If you vary the immersion depth of the stopper (arrow in picture no.6) you can change the gas pressure so the motor keeps running. |
|
T 1 |
Make a sample collection bag for each sample you plan to take. |
|
T 2 |
Take your samples, number each of them and match them with the criteria of table no.1:
(The samples should be analysed at the same day.) |
|
Table 1 |
without exhaust catalyst |
with exhaust catalyst |
|
. |
cold engine |
engine at operating temperature |
cold engine |
engine at operating temperature |
|
petrol engine |
4 stroke |
. |
. |
. |
. |
|
2 stroke |
. |
. |
. |
. |
|
diesel engine |
4 stroke |
. |
. |
. |
. |
|
(2 stroke) |
. |
. |
. |
. |
|
3. Qualitative analysis of exhaust gas (CO, CO2 und NOx)
|
T 3 |
Read the information below . Take a sheet of paper and write down the missing reaction schemes! |
Information on the identifications:
1. Identification of carbon monoxide with an ammoniac solution of silver nitrate:
Properties of carbon monoxide: solubility in water 0,23g/L (20°C), bp: -191°C, reducing agent, highly toxic in concentrations w>5%, MAK: 30ppm
Reaction scheme for this identification:
2. Identification of carbon dioxide with lime water (aqueous solution ofcalcium hydroxide):
Properties of carbon dioxide: solubility in water 1,78g/L (20°C), subl.: -79°C, MAK: 5000ppm
Reaction scheme for this identification:
3. Identification of nitrogen oxides with Saltzmann solution:
Properties of nitrogen dioxide: reacts with water yielding HNO2 and HNO3, bp:21°C, vapour pressure: 1013hPa at 20°C, highly toxic in concentrations w>1%, MAK: 5ppm
Reaction scheme for this identification: |
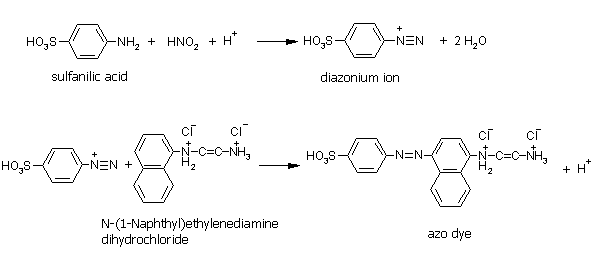 |
 |
|
Picture 7: The reaction scheme of the nitrogen identification with Saltzmann solution.
|
|
Exhaust gases contain substances that are hazardous to your health:
When carrying out the experiments use a fume hood or work outside !
|
T 4 |
Plan an experimental set-up and a procedure that meet the following criteria:
- Each exhaust gas sample should be simultanuously tested for carbon monoxide, carbon dioxide and nitrogen oxides.
- The identification should be arranged in an order in which they do not falsify of interfere with each other. (Take into consideration the different solubilities, reactivities with water and reaction products.)
- A semi-quanitative differentiation of the concentration of the pollutants in the different exhaust gas samples should be made by controlling the volume of the tested exhaust gases.
|
|
T 5 |
Carry out your analysis of the exhaust gas samples and list your results in a table. |
|
About this page:
Authors: M. Seesing, M. Tausch - Universität Duisburg-Essen, Duisburg / Germany
Rewiewer:
Last update: 2004-05-24 |
|
 |
|









