|
|
 |
|
|
|
 |
 |
|
|
 |
Context: The nitrogen cycle
|
Key words:
Nitrogen fixation, Nitrification, Denitrification, Assimilation, Fertilisation, Nitrogen oxide emissions | |
|
 The nitrogen cycle The nitrogen cycle
The nitrogen cycle is essential for the life on planet Earth and certainly also for our life as humans. Nitrogen compounds play an important role in the metabolism of living organisms. Many organic molecules in the bodies of plants, animals and humans include nitrogen compounds (for example amino-acids, proteins and DNA).
But nitrogen compounds are also present in chemical processes in the atmosphere and have an impact on the climate system.
|
 |
 |
|
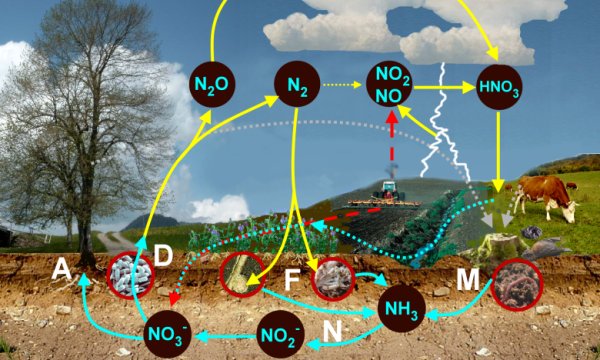
1. The nitrogen cycle: A = Assimilation by plants, F = Fixation of nitrogen by bacteria either in symbiosis with plants or by free living bacteria in the soil, N = Nitrification, D = Denitrification, M = Mineralisation - Please click the image in order to enlarge it. (140 KB)
Scheme: Elmar Uherek
Basically the nitrogen cycle consists of chemical reactions in the air (oxidation is dominant) and of chemical reactions in the biosphere, either plants or microorganisms in the soil (oxidation or reduction).
|
 Nitrogen fixation, nitrification and denitrification Nitrogen fixation, nitrification and denitrification
Plants depend on nitrogen compounds in order to grow. In nature, nitrogen can be available in forms accessible to plants (like nitrates or ammonia compounds) or inaccessible to plants (like molecular nitrogen or nitrous oxide). The conversion between both forms takes place during nitrogen fixation or denitrification.
Nitrogen fixation is the process of making available nitrogen compounds in the air (mainly molecular nitrogen N2) to plants. Nitrogen fixation is possible for many bacteria and cyanobacteria. They live either in the soil or in association with plants or also (few) animal species. For example, the legume family of plants (Fabaceae) contains such bacteria in their roots.
Products of nitrogen fixation are ammonia, nitrites or nitrates.
Nitrification is the second step of nitrogen fixation. If we see ammonia as primary product of nitrogen fixation we can call the conversion of ammonia to nitrites and nitrates (carried out by nitrifying bacteria) nitrification.
|
 |
 |
 |
|
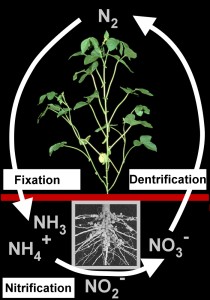
2. The biological nitrogen cycle in the soil consists of a) nitrogen fixation by bacteria which can live in symbiosis with plants (like here with the roots of the soy bean) and b) denitrification by other bacteria which set free molecular nitrogen or N2O in nitrate respiration. Please click the scheme to enlarge it. (50 KB)
Collage: Elmar Uherek
|
|
|
Denitrification is the process of reduction of nitrate to nitrogen. It is carried out by microorganisms (Bacillus, Pseudomonas and other bacteria) which switch from oxygen to nitrate respiration in particular in insufficiently ventilated soils. The denitrification process can lead to several intermediates. The most relevant is nitrous oxide. The gases go back to the atmosphere. Nitrous oxide is a long-lived greenhouse gas.
Nitrogen fixation is the opposite process of denitrification.
|
 |
 |
|
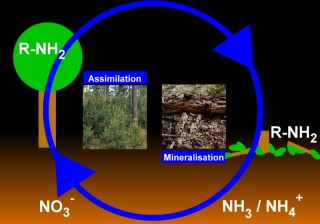
3. Assimilation and mineralisation determine the uptake of nitrogen compounds from soil and the incorporation into the biomolecules of plants, or the conversion to anorganic nitrogen after the death of the plant respectively. Please click on the image for a larger view. (70 KB)
Scheme: Elmar Uherek
|
|
 |
 Assimilation and mineralisation Assimilation and mineralisation
Accessible nitrogen compounds can be stored in the soil in an inorganic form (nitrate) or be included into a living organism as organic nitrogen.
Assimilation is the conversion of inorganic nitrogen (such as nitrate) into an organic form of nitrogen like, for example, an amino acid. Nitrate is reduced for this purpose by enzymes first to nitrite (by nitrate reductase), then to ammonia (by nitrite reductase). Ammonia is incorporated into amino acids.
|
Mineralisation (ammoniafication) is a process during which decomposers like earthworms, termites, slugs, snails, bacteria or fungi convert the organic nitrogen of dead plants into inorganic forms. The first step is formation of ammonia and its salts (NH4+X-).
Assimiliation and mineralisation are opposed processes
|
 |
 |
 |
|
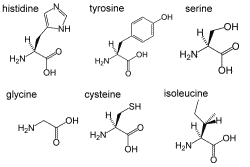
4. Amino acids (here a selection) are nitrogen containing biomolecules. Porteins are built from amino acids.
|
|
|
 Atmospheric oxidation Atmospheric oxidation
The most important pathways of natural production of nitrogen oxides are oxidation processes under very hot conditions. This can be combustion in the hot part of vegetation fires or oxidation of the (under normal conditions inert) molecular nitrogen in lightning or volcano eruptions.
The main products of such processes are nitrogen oxide NO and nitrogen dioxide NO2 and after further reaction nitric acid. As explained in the research text, certain concentrations of NO and NO2 lead to ozone formation, if supported by sunlight and emissions of hydrocarbons.
|
 |
 |
|
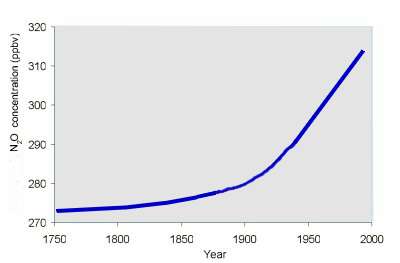
5. The increasing fraction of dinitrogenmonoxide (laughing gas) in air is first of all a consequence of nitrogen fertilisation. Diagramm: Elmar Uherek
|
|
 |
 Human influence Human influence
Nitrogen oxides and ozone smog
In addition to natural sources, humans are responsible for emissions of NO / NO2 from technical combustion processes. The most dominant one is the combustion taking place in car engines. If average concentrations in the air become too high this can, under certain conditions, lead to ozone smog.
Furthermore, nitrogen oxides are converted in atmospheric reactions into nitric acid. This contributes to acid rain and its negative consequences.
|
Fertilisation and eutrophication
In order to improve growing conditions for crops farmers bring nitrogen containing fertilisers into the soil. This could be called a technical nitrogen fixation, based for example on the ammonia synthesis in the Haber-Bosch process.
|
 |
|
 |
 |
|
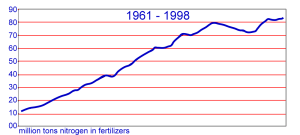
7. Nitrogen containing fertilisers have been produced in strongly increasing amounts over recent decades. Graph adopted from IFA.
|
|
 |
If too much fertiliser is added and not taken up by plants it causes a twofold negative impact. Ammonia and nitrate are washed out and end up in water bodies and coastal areas. Here they cause strong growth of plants and algae (phytoplankton) and as a result life in the water can be extinguished if the oxygen content is too much reduced due to decomposition of dying plants.
|
|
Moreover denitrification is enhanced and more nitrous oxide N2O is produced. This increases the greenhouse effect and the nitrous oxide reaching the stratosphere is converted into other nitrogen oxides fostering the depletion of the ozone layer.
|
Author:
Elmar Uherek - Max Planck Institute for Chemistry, Mainz
|
|
 |
|







