|
|
 |
|
|
|
 |
| |
|
|
 |
Higher Atmosphere
Basics |
Measurements in the Stratosphere
When we talk about concentrations of chemicals in the statosphere, the question may arise, how we know about such compounds and their appearance. The stratosphere begins in about 8-15 km of altitude and the interesting regions are higher than normal planes go.
|
|
|
|
|
 |
|
There are two possibilities to measure compounds in the stratosphere.
- Instruments can be carried to the stratosphere by special aircrafts or balloons.
- The interaction of light with molecules in the air can be used to study the stratosphere from the ground or from space (satellites).
|
Air Planes
Unique measurements have been made possible with special air planes, like the former Russian high altitude spy plane, which is now called "Geophysica". It has been converted into an airborne laboratory. Such planes reach altitudes of about 20 km. But the flights are very expensive.
|
 |
 |
 |
|
1. Geophysica - high altitude research aircraft
source: MDB Design Bureau
|
|
Balloons
A more common alternative are balloon measurements. Weather balloons can reach 30-35 km before they burst and e.g. carry a little ozone sensor. A chemical reaction takes place in the sensor and tells, how much ozone is in the air. The information is sent via a radio signal to the Earth. Although ozone is nowadays measured by satellites, balloons are still better for vertical profiles.
|
 |
 |
|
2. a) Start of an ozone balloon at Hohenpeissenberg Observatory
courtesy of Ulf Köhler
Please click to enlarge! (85 K)
|
|
 |
 |
 |
|
2. b) Ozone probe for balloon measurements
courtesy of Ulf Köhler, DWD Hohenpeissenberg
Please click to see details! (80 K)
|
|
Light interaction
Interaction phenomena of light and molecules are difficult to understand and would require some knowledge in quantum physics, if we would really try to explain them. However, we have to keep in mind: Something happens if light and matter interact. Light can be taken up, reflected, scattered or taken up and re-emitted as another sort of light (in another wavelength regime).
|
The direct sunlight is blocked by the clouds, if we dive in the deep sea it becomes darker and darker because more and more light is lost and also a dust storm in the desert makes the sun pale. Not only big particles and clouds and water absorb or reflect light, also the smaller molecules. They can scatter the light back to the Earth or change their state and emit less energetic light of another wave length. We know this phenomena (fluorescence or phosphorescence) from some toys, which take up daylight or room light and emit different light in the dark. The sort of the light tells us something about the character of the compounds. The intensity of the light tells us something about their concentration.
|
 |
 |
 |
|
3. Phosphorescence takes place, if light is absorbed and reemitted again at an other wavelength.
source: composed from web-advertisements
|
|
 |  | 4. How does a LIDAR work?
Please press reload in order to restart the animation!
by EU
|
|  | Interactions of light with molecules in the stratosphere can be observed from the ground or be measured from the space on satellites.
Lidar
Lidar (light detection and ranging) is one technique, which can be used from the ground. A short laser pulse of very intensive light is sent to the sky. After a while the scattered or re-emitted light comes back and is measured. We get information which compounds we find (wavelength of the returning light) and in which concentration (intensity of the returning light).
But from which altitude does the light come back? From 10km? 30 km? Light has a certain velocity. The longer it takes after the laser pulse, the higher are the molecules, where the light comes from.
|  |  |  | 5. LIDAR measurements
Image source: University of Western Ontario
The animation on the left shows a laser pulse (light blue) whose light is scattered back in three different altitudes by air molecules (green) and arrives at the detector (light green) at three different times.
|
|
| | | |  | |
 |
 |
|
6. SODAR - wind speed measurements
image source: Meteotest
|
|
 |
RADAR and SODAR
Different variants of wave detection and ranging technique are used, e.g. with infrared light. Better known is RADAR (Radio detection and ranging), which is used to measure particles in the air and the properties of clouds. RADAR allows to track thunderstorms over several hundred kilometers. If sound is used instead of light (SODAR = sound detection and ranging) we have a powerful tool for the measurements of wind speed and direction.
Satellites
Satellites are observing our planet from the space. Some of them remain always over the same part of the Earth (geostationary satellites), some are running at an altitude of 500 - 1000 km and surround the Earth in about 1.5 to 2 hours. On a few satellites there are spectrometers installed, i.e. instruments which detect light. The light has gone through the atmosphere and interacted with the molecules. Different sorts of measurements are possible for the purpose of atmospheric research.
|
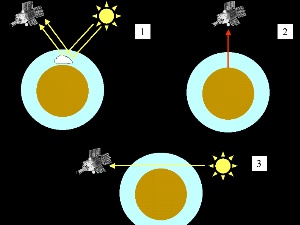
The satellite may measure sunlight which is scattered back by the clouds or the air molecules (1). Infrared spectrometer can also measure the long wave radiation with comes directly from the Earth (2). For certain positions of sun, Earth and the satellite, the sun beams pass the Earth tangentially and go directly through the atmosphere to the detector on the satellite (3). Depending on the angle the light passes through different parts of the atmosphere and altitude related information can be gained.
|
 |
 |
 |
|
7. Different techniques of satellite measurements
scheme by Elmar Uherek
Please click to enlarge! (60 K)
|
|
About this page:
author: Dr. Elmar Uherek - Max Planck Institute, Mainz
scientific reviewer: Dr. John Crowley, Max Planck Institute for Chemistry, Mainz - 2004-05-04
educational proofreading: Michael Seesing - Uni Duisburg - 2003-07-02
last published: 2004-05-05
|
|
 |
|









