|
|
 |
|
|
|
 |
| |
|
|
 |
Lower Atmosphere
Read more |
Fire chemistry and global importance
Every year large areas of our planet burn:
10 Mio ha of boreal forest in the northern latitudes
40 Mio ha of tropic and sub-tropic forest
500 - 1000 Mio ha of open forest and savannahs
1 Mio ha = 1 Mio x 100 m x 100 m = 10,000 km2
Imagine the area:
|
|
|
|
|
 |
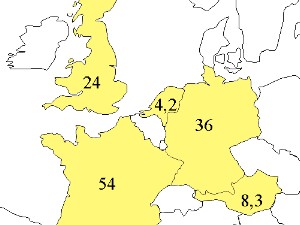 |  | 1. Area of European countries in Mio ha,
e.g. Netherlands = 41.500 km2
image: Elmar Uherek |
|  |  |  | 2. Smoke over Siberia
Image: courtesy the SeaWiFS Project
Due to the focus on the rainforests people often forget the huge boreal forests e.g. in Siberia (photo) and Canada. They cover an area of 920 Mio ha of closed mainly coniferous forest, make up for 73% of the global coniferous forest area and for 29% of the worlds forest. Nowadays boreal forest fires are not only caused by nature but also by humans.
|
|
| |  | |
|
The global carbon budget of CO2 exchange for the period 1850 - 1990
in Pg Carbon (Pg C). 1 Pg = 1015 g = 1 billion tons
Emissions from man made fires are an important source. |
 |
 |
|
3. Carbon dioxide - sources and sinks - image: Elmar Uherek
|
|
The main sources are:
1) landuse change (+ 121 Pg C)
2) fossil fuel burning (+ 212 Pg C)
|
 |
The main sinks are:
3) CO2 uptake by the oceans (- 107 Pg C)
4) CO2 uptake by the continents
(- 82 Pg C)
|
|
The CO2 uptake by the continents is poorly understood. Probably it is primarily due to increasing plant growth at other places than the typical burning regions.
90% of the CO2 emissions from landuse change are from deforestation. A major problem in the rainforests is, that once burnt down a secondary vegetation grows, which burns yearly. This prevents a reforestation.
Natural vegetation fires can be short term sources, but in the long term they level out or even add up to a very small sink (< 0.1 Pg C / year), since carbon sediments are stored in the ground. Open forest and savannah fires occur in cycles of 1-3 years on much larger areas than rainforest fires without major perturbation of the climate system.
|
 |
 |
|
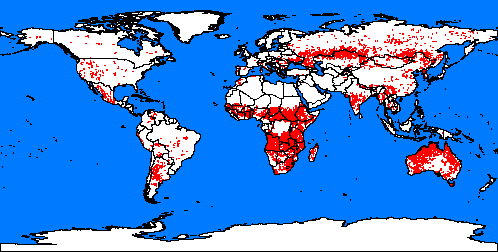
4. Fires spots around the word
Map: from Global fire monitoring centre
|
What is formed during the combustion process?
This depends on
a) of what compounds the plants consist
b) on the temperature of the flames
c) if enough oxygen is available
Is it an open fire or a smouldering fire?
What do plants consist of?
The water content of plants can be up to 60%, but usually before the fire seasons the vegetation looses a lot of water during drought times. A typical composition of the organic dry matter is:
elemental:
carbon (C): 45%
oxygen (O): 42%
hydrogen (H): 6%
others (N, S, P): 7%
e.g. sulphur 0,1-0,5%
components:
- cellulose and hemicellulose [carbohydrates (C6H10O5)n]: 50-70%
- lignin: 15-35%
- smaller amounts of: proteins, amino acids, other metabolites
- minerals: up to 10%
|
 |
 |
 |
|
5. The picture shows the same place of a forest fire two years later after reseeding and rehabilitation efforts.
photo: Dylan Rader (full size: 75 K)
 wildland fire pictures wildland fire pictures
|
|
 |
 |
|
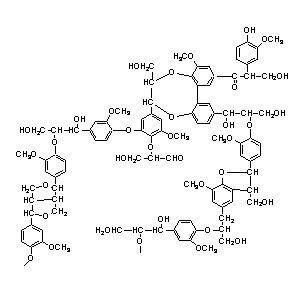
6. Lignin gives footing to the wood.
|
|
 |
Lignin is formed by the irreversible removal of water from sugars to create aromatic structures. The OH groups can react with each other or with the aldehyde or ketone groups. The result is a polymeric structure with ether bonds.
The burning process:
After the ignition a period of open fire (sufficient oxygen supplied) is usually followed by a period of smouldering fire (not enough oxygen available).
In an open fire a simple mixture of a few fully oxidised compounds is formed. In smouldering fire a complex mixture of many not fully oxidised compounds is formed, sometimes even methane (CH4).
|
|
After ignition (> 180°C / 450 K) flaming combustion (> 580°C / 850 K) starts: Simple molecules as CO2, H2O, NO, N2O, N2 and SO2 are released as oxidation products. After that the flames cease ...
|
 |
... and smouldering (< 580°C / 850 K) begins. Due to a lack of oxygen CO and many partially oxygenated organic compounds (formaldehyde, acetaldehyde, methanol, acetone, methane) are released.
|
About this page:
author: Dr. Elmar Uherek - Max Planck Institute for Chemistry, Mainz
scientific reviewer: Dr. Rolf Sander - Max Planck Institute for Chemistry, Mainz 2004-05-18
last published: 2004-04-06 |
|
 |
|









